

APM 777
World’s first blend of APP + APX non toxignic toxins + M.hyo bacterin
- Origin: Nisseiken Co., Ltd. (Japan)
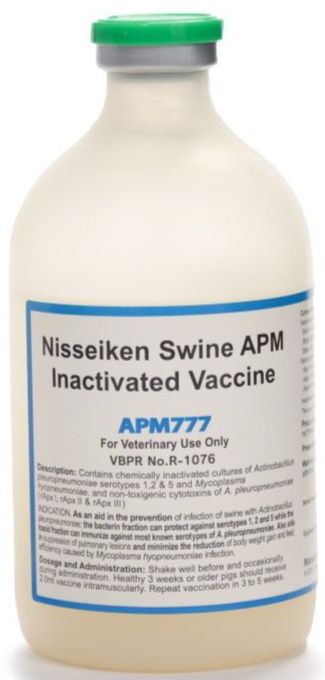
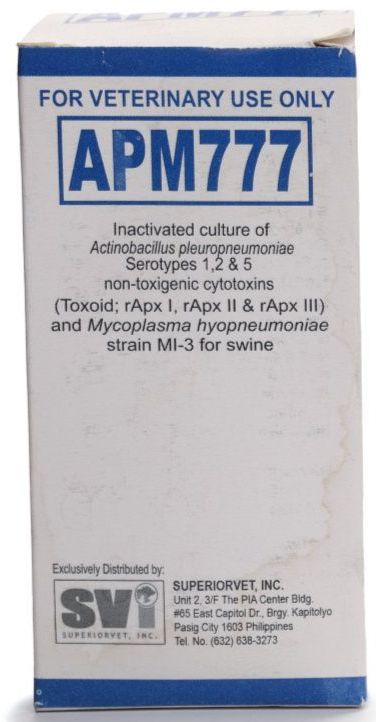
- Active Ingredient: Inactivated APP Serotype 1,2,5, rAPX I,II,III, Inactivated Mycoplasma hyopneumoniae strain MI-3
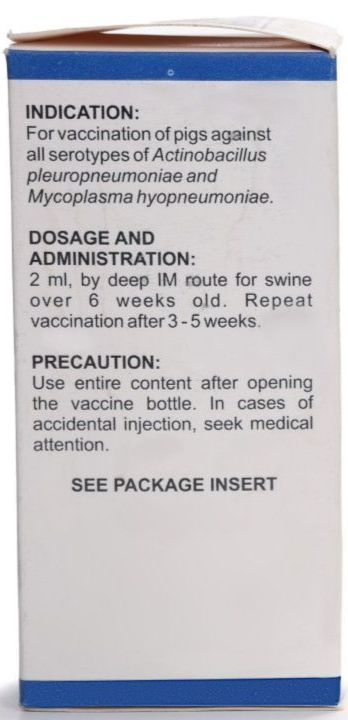
- Dosage and Administration: Inject 2-mL vaccine intramuscularly into swine (3 weeks old or over). Repeat vaccination after 3-5 weeks from initial administration
- Presentation: 1 bottle contains 100mL (50 doses)
- Storage: Store at 2°C to 10°C. Use the entire content after opening the vaccine bottle.
